Atomic Theory

The theory that all matter is made up of tiny indivisible particles (atoms). According to the modern version, the atoms of each element are effectively identical, but differ from those of other elements, and unite to form compounds in fixed proportions.
Nucleus

The positively charged central core of an atom, containing most the atoms mass.
Proton

A stable subatomic particle occuring in all atomic nuclei, with a postitive electric charge in magnitude to that of an electron, but opposite of sign. We know how many there are in a particle by the atomic number. Mass= 1.67262158x 10^-27 kilograms
Electon

A stable subatomic particle with a charge of negitive electricty, found in orbitals in all atoms and acting as the primary carrier of electricity in solids. We know how many there are in a particle by the atomic number. Mass= 9.10938188x 10^-31 kilograms
Neutron
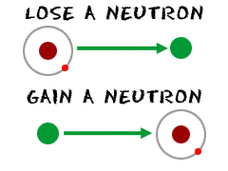
A subatomic particle of about the same mass as a proton but without an electical charge, present in all atomic nuclei except those of ordinary hydrogen. We know how many by the atomic mass minus the amount of protons.
Mass= 1.6749x 10^-27
Mass= 1.6749x 10^-27
Orbital
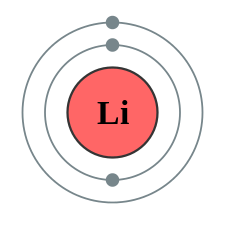
Each of the actual or potential patterns of electron density that may be formed in an atom or molecule by one or more electrons, and that can be represented as a wave function.
Energy Level
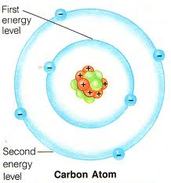
The fixed amount of energy that a system described by quantum mechanics, such as a molecule, atom, electron, or nucleus, can have.
Valence Electron
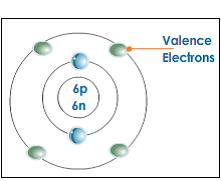
An electron in the outer shell of an atom which can combine with other atoms to form molecules.
Isotope
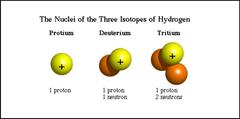
A particular form of an element. The number of protons and electrons are constant but the number of
neutrons change. An atom becomes this when it gains or loses neutrons. The average of all isotopes masses that can be found naturally of an element is called the Average Atomic Mass or AMU.
neutrons change. An atom becomes this when it gains or loses neutrons. The average of all isotopes masses that can be found naturally of an element is called the Average Atomic Mass or AMU.
Ion
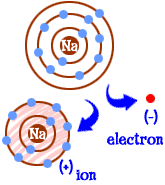
An atom that has lost or gained an electron and is now charged.
Strong Nuclear Force
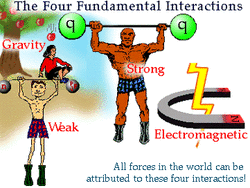
A fundamental force that is associated with the strong bonds created by subatomic particles.
